Cutting paper into tiny pieces or crushing a rock are obvious physical changes but to restore the original piece of paper or rock is difficult. Many junior school science texts state that chemical changes are irreversible while physical changes can be reversed. Most chemical reactions are reversible although this can be difficult in practice. Many examples of materials dissolving (for example, an Alka Seltzer in water, metal in acid and the effect of acid rain on marble and concrete) involve both chemical and physical processes.
Dissolving of instant coffee in water seems to be a physical change but in most cases dissolving is accompanied by an energy change and is probably better considered to be a chemical process even though it is possible to recover the original components by physical means. For example salt dissolving in water is usually considered to be a physical change, however the chemical species in salt solution (hydrated sodium and chlorine ions) are different from the species in solid salt. In fact this should be considered more of a continuum. Frequently students are led to believe that a change is either physical or chemical.
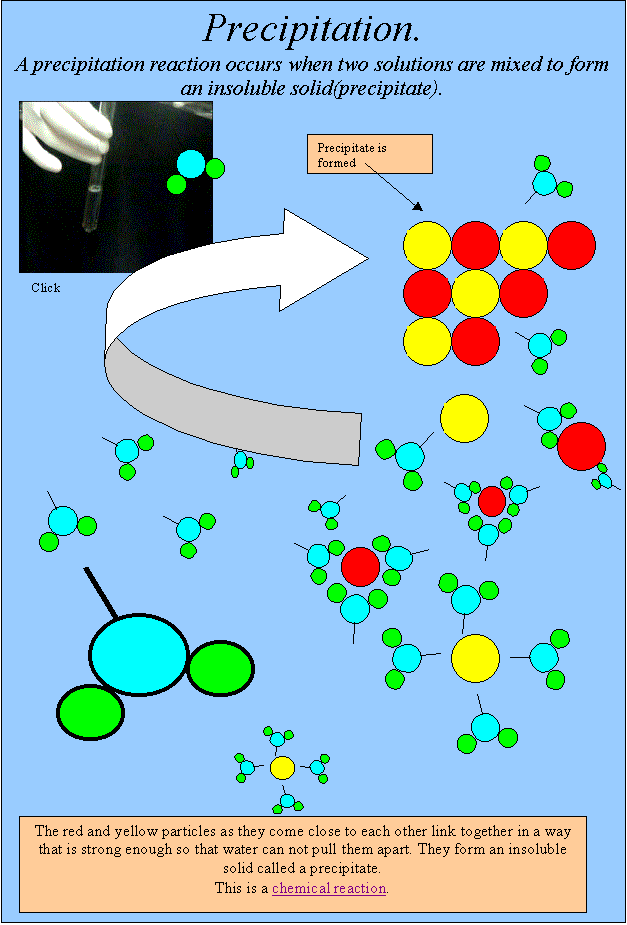
The distinction between physical and chemical change is not clear cut. However in a chemical change, the kind of matter changes and at least one new substance with new properties is formed. In a physical change the appearance or form of the matter changes but the kind of matter in the substance does not. Other commonly held views are that chemical changes are caused by the mixing of substances/reactants or that heat (which is considered to be some form of material) has to be added. A common everyday application of a reversible chemical reaction is the charging and discharging of rechargeable batteries – including car batteries however students may believe that batteries are a container of stored electricity, rather than of chemicals that react in ways that convert chemical energy into electrical energy. Students also frequently believe that the original substance in a chemical reaction vanishes completely and forever. Students commonly believe that physical changes are reversible while chemical changes are not. Students may believe that beer frothing is an example of a chemical change or an apple ripening is a physical change. Many students did not appreciate that a chemical change is characterised by the formation of a substance having different properties from the original substance and a considerable proportion of students who did were unable to offer suitable reasons for distinguishing a ‘new’ substance. Research: Driver, Rushworth & Wood-Robinson (1994) Melting and expansion on heating were also considered to be evidence of chemical change by some students. Students can consider potassium permanganate (Condy's crystals) dissolving in water to be a chemical change because of the intense difference in colour. One study found that 80% of students considered a difference in colour between the reactant and product evidence of chemical change. If students regard ice as a different substance from liquid water they are likely to classify the melting of ice as a chemical change.

This depends on their conception of substance. Freezing and boiling are considered to be examples of chemical reactions. Research shows that students frequently use the term chemical change to describe changes in physical state. Research: Loughran, Mulhall & Berry (2002) Pat (recording the group's notes about a prac): What happened? However an understanding of the differences between purely physical processes such as melting, evaporation and boiling and the changes that take place in chemical reactions, particularly the idea that new substances are formed, is important to an understanding of chemistry and students very frequently confuse the two. Students have difficulty distinguishing between physical and chemical change, despite formal teaching, and the distinction is somewhat arbitrary. Contrasting student and scientific views.
